Formlabs BioMed Durable Resin 1L (Form 4)
- Home
- Shop
- เรซิ่น, เรซิ่น วิศวกรรม, เรซิ่น ทางการแพทย์
- Formlabs BioMed Durable Resin 1L (Form 4)




Formlabs BioMed Durable Resin 1L (Form 4)
ความเข้ากันได้ของเครื่องพิมพ์: Form 4B, Form 4BL
เข้ากันได้กับถังเรซินเหล่านี้: Form 4 Resin Tank, Form 4L Resin Tank
เข้ากันได้กับแพลตฟอร์มการพิมพ์เหล่านี้: Form 4 Build Platform, Form 4 Build Platform Flex, Form 4L Build Platform L
ได้รับการรับรองว่าเป็นชีวภาพ (Certified Biocompatible).
- คำอธิบาย
คำอธิบาย
This post is also available in: English (อังกฤษ) 日本語 (ญี่ปุ่น)
BioMed Durable เรซิ่น (Form 4) – เรซิ่นทนทานทางการแพทย์
เรซิ่นทนทานที่ปลอดภัยสำหรับเครื่องพิมพ์ Form 4
BioMed Durable เรซิ่น (Form 4)) เรซิ่นที่ออกแบบมาสำหรับเครื่องพิมพ์ Form 4 ให้ความทนทานสูงและปลอดภัยต่อร่างกาย เหมาะสำหรับการสร้างแบบจำลองและอุปกรณ์ทางการแพทย์
DOCUMENT TDS.PDF
BioMed Durable Resin
คำอธิบายผลิตภัณฑ์
BioMed Durable Resin เป็นวัสดุการพิมพ์ 3D ที่โปร่งใสสำหรับการใช้งานที่ต้องการความเข้ากันได้ทางชีวภาพซึ่งต้องการความต้านทานต่อการกระแทก การแตกหัก และการขีดข่วน วัสดุนี้ได้รับการรับรองตามมาตรฐาน USP Class VI และสามารถใช้ในแอปพลิเคชันที่ต้องการการสัมผัสกับผิวหนังในระยะยาว (>30 วัน) และเยื่อบุผิว (>30 ชั่วโมง) หรือการสัมผัสกับเนื้อเยื่อ กระดูก และเนื้อเยื่อฟันในระยะสั้น (<24 ชั่วโมง)
BioMed Durable Resin ผลิตขึ้นในสถานที่ที่ได้รับการลงทะเบียนจาก FDA และได้รับการรับรอง ISO 13485 วัสดุนี้ยังได้รับการรับรอง USP Class VI ทำให้เหมาะสมสำหรับการใช้งานในด้านเภสัชกรรมและการส่งยา
ทำไมจึงควรเลือก BioMed Durable Resin?
ใช้การพิมพ์ 3D และ BioMed Durable Resin เพื่อผลิตอุปกรณ์ที่ใช้งานจริงซึ่งมีความสำคัญต่อประสิทธิภาพ ความต้านทานต่อแรงกระแทก และความเข้ากันได้กับชีวภาพ
- ทนต่อแรงกระแทก แตกหัก และการเสียดสี
ผลิตชิ้นส่วนที่มีคุณภาพทางการแพทย์ที่ทนต่อแรงกระแทก โดยมีค่า Notched Izod เท่ากับ 98 J/m - วัสดุที่ได้รับการรับรองในระดับการแพทย์
ใช้วัสดุที่ผลิตภายใต้ระบบการจัดการคุณภาพของเรา โดยมีความมุ่งมั่นในการปฏิบัติตามมาตรฐาน ISO 13485 อย่างเคร่งครัด - ชิ้นส่วนที่เข้ากันได้กับชีวภาพ
การทดสอบกรณีการใช้งานทั่วไปโดย Formlabs มีจุดประสงค์เพื่อสร้างความมั่นใจว่ามาตรฐานนี้เหมาะสมสำหรับการใช้งานทางชีวภาพ - ความใสและพื้นผิวที่ยอดเยี่ยม
ชิ้นส่วนที่ผลิตด้วยวัสดุนี้ดูและรู้สึกเหมือนชิ้นส่วนที่ใช้งานจริง และมีพื้นผิวที่เรียบเนียนมาก

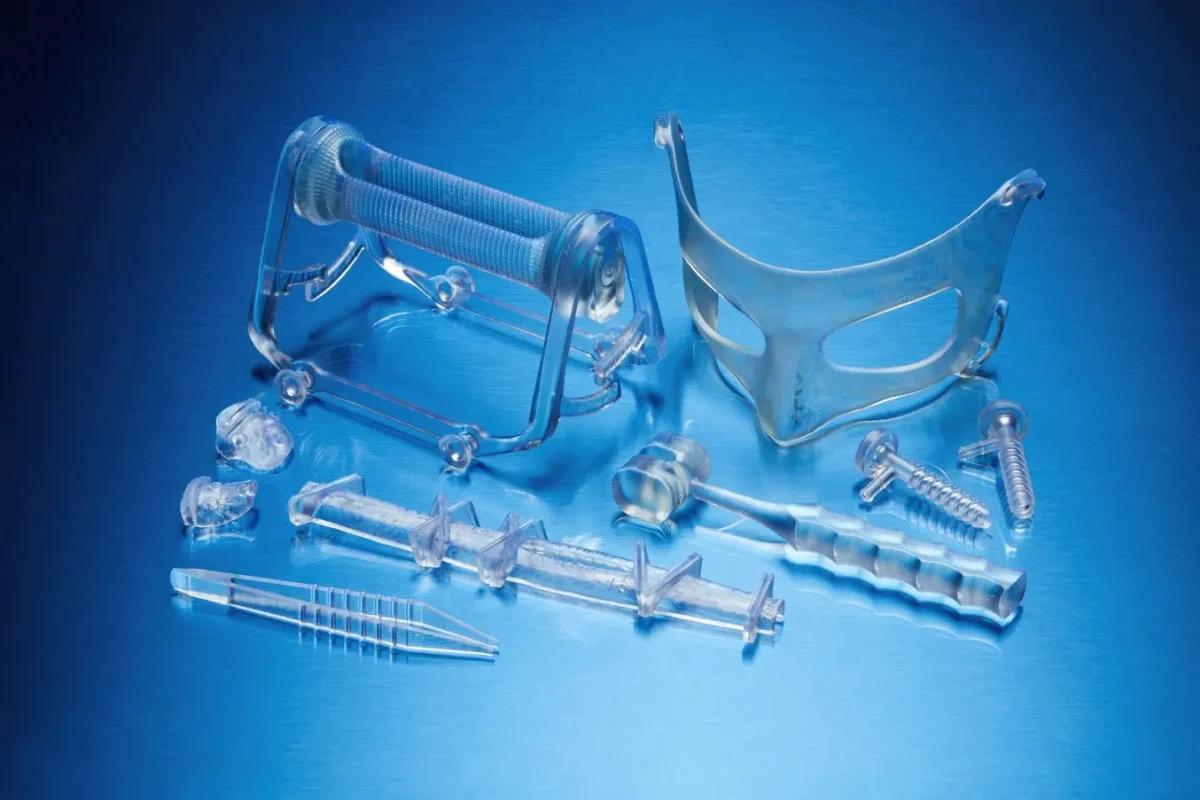
การใช้งาน *
นำเสนอต้นแบบเครื่องมือเฉพาะผู้ป่วย (PSI) และอุปกรณ์ทางการแพทย์ที่เฉพาะเจาะจงกับกระบวนการที่จุดดูแลผู้ป่วย ปรับปรุงผลลัพธ์ของผู้ป่วย ลดภาวะแทรกซ้อน และลดเวลาในการดำเนินการด้วยอุปกรณ์ทางการแพทย์ที่พิมพ์จาก BioMed Durable Resin
BioMed Durable Resin เหมาะสำหรับ:
- อุปกรณ์เฉพาะบุคคลสำหรับผู้ป่วย
- อุปกรณ์ใช้แล้วครั้งเดียว
- อุปกรณ์และชิ้นส่วนที่ต้องการความเข้ากันได้กับชีวภาพและความต้านทานต่อแรงกระแทก
Material Properties*
| คุณสมบัติ | ค่าคุณสมบัติ |
|---|---|
| ความต้านทานแรงดึงสูงสุด | 29.1 MPa |
| โมดูลัสแรงดึง | 994 MPa |
| การยืดตัวที่จุดแตก | 33% |
| ความต้านทานแรงดัด | 21 MPa |
| โมดูลัสของแรงดัด | 643 MPa |
| ความแข็ง (Shore D) | 75 D |
| ค่าความต้านทานการกระแทกแบบ Notched Izod | 98 J/m |
| อุณหภูมิการเบี่ยงเบนความร้อน @ 0.45 MPa | 46 ºC |